Authors: Leblans NIW1, Stuart-Haëntjens E2, Schindlbacher A3, Vicca S1
Reviewer: Almagro M4
Measurement unit: Mg C ha-1 or kg C m-2; Measurement scale: plot; Equipment costs: €; Running costs: €; Installation effort: medium; Maintenance effort: -; Knowledge need: medium; Measurement mode: manual
The soil carbon (C) stock is the amount of C stored in the soil (Davidson & Janssens, 2006) and is the product of the long-term balance between C inputs from aboveground and belowground net productivity and C outputs from decomposition and erosion (Kirschbaum, 1995; Melillo et al., 2002). Soil C stocks are sensitive to climate change, because virtually all processes that regulate C inputs and outputs are driven by climate-related factors (such as temperature, CO2 concentration, rainfall, moisture and nutrient availability) (Tian et al., 2015). Factors other than climate that strongly influence soil C stocks are land-use (Guo & Gifford, 2002) and disturbances, such as erosion (Panagos et al., 2015), volcanic eruptions (Gísladóttir et al., 2010), fire, and human and animal activities (Laurel & Wohl, 2019). The sensitivity of soil C stocks to climatic changes implies that the C sink or source capacity of soils can drastically shift under altering climatic conditions (Kirschbaum, 1995; Melillo et al., 2002; Davidson & Janssens, 2006). Since soils store globally three times more C than the atmosphere (Chapin et al., 2011), these shifts could, in turn, induce powerful feedbacks to the climate system (Kirschbaum, 1995; Melillo et al., 2002; Davidson & Janssens, 2006; Scharlemann et al., 2014; Tian et al., 2015). This is why a detailed quantification of the current global soil C stocks and potential shifts under land-use and climate change are important.
The soil nutrient stock is the amount of nutrients stored in the soil (Chapin et al., 2011), with a focus on nitrogen (N), phosphorus (P), and potassium (K), as these elements are most frequently limiting for plant growth (Sterner & Elser, 2002). Soil nutrient stocks have strong associations with soil C stocks, as nutrients and C are tightly coupled in soil organic matter and are affected by the same processes (Hobbie et al., 2002). Furthermore, plant nutrient availability, which is often linked to soil nutrient stocks (Saynes et al., 2005), has a strong impact on plant growth and litter quality and thus indirectly on the soil C balance (Li et al., 2011).
2.2.4.1 What and how to measure?
Soil C and nutrient stocks are commonly assessed through destructive soil sampling and subsequent determination of the C and nutrient concentration in the soil (excluding particles > 2 mm) (Jobbágy & Jackson, 2000). The sampling is generally performed using manual soil augers. These can differ in type, depending on the soil texture, but have in common that the exact core volume must be known. Augers that strongly disturb the soil volume are thus not suitable for this measurement.
Measurements of soil C are mostly done by dry combustion (Matejovic, 1997; Senesi & Senesi, 2016). Other techniques have also been used in the past (most commonly the Walkley‐Black method (wet acidified dichromate oxidation) or loss on ignition), and conversion factors for comparison can be found in Soon & Abboud (1991). A new, less laborious method for laboratory and field determination of soil C, i.e. laser-induced breakdown spectroscopy (LIBS), is currently being developed (Senesi & Senesi, 2016). Methods used for soil nutrient determination depend on the nutrient under investigation. Nitrogen is generally measured together with C using dry combustion (Matejovic, 1997). The other most commonly measured nutrients (P and K) are measured by acid destruction with H2SO4, salicylic acid, H2O2, and Se (Wallinga et al., 1989). Other important elements (Ca, K, Mg, Na, Mn) can be determined with the same method.
Installation, field operation, maintenance, interpretation
At the sampling spot, the vegetation and the superficial litter layer are removed down to the topsoil layer (Vadeboncoeur et al., 2012). Typically, one core is taken per plot, but in heterogeneous environments it is more desirable to collect a number of smaller cores that are subsequently pooled before analyses and provide a more accurate characterisation of the plot. The latter technique (with more but smaller volumes) yields larger uncertainty in the core volume, and thus in the transformed C and nutrient content per unit area measurements.
To determine the soil carbon stock per unit area, the entire soil depth needs to be sampled. A core is taken down to the bedrock and is split up into different depth layers to obtain information about the depth profile of the stocks (Maillard et al., 2017). In general, a subdivision is made between topsoil (0–10 cm depth) and subsoil (> 10 cm depth), but in some cases, it might be important to use a finer scale. Alternatively, soil horizons are used as subdivisions instead of fixed soil depth (Maaroufi et al., 2015). For more information on the importance of sampling design, the spatial scale of the sampling, and sampling depth, one can consult Maillard et al. (2017) and Allen et al. (2010).
Soil samples are sieved at 2 mm to exclude stones and roots and dry weight is determined (optimally the samples are dried at a maximum of 40 °C to avoid volatilisation of N). Subsequently, the C and nutrient content of finely ground aliquots is determined (Matejovic, 1997). Finally, the C and nutrient stocks per unit area can then be calculated by applying the following equation:
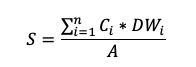
where S is the C or nutrient stock over the entire soil depth, which is split into n layers. Ci is the C or nutrient concentration for layer i (expressed in%), DWi is the dry weight of the core taken from layer i, and A is the surface area of the core.
Alternatively, the C and nutrient stock can be expressed per volume instead of per area. This is necessary when it is not desirable or possible to sample the entire soil depth. In that case, the C or nutrient stock of the soil layer of interest (Si) is calculated using the volume of the sampled core (Vi):
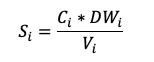
Where to start
Davidson & Janssens (2006), Jobbágy & Jackson (2000), Maillard et al. (2017), Melillo et al. (2002), Senesi & Senesi (2016)
2.2.4.2 Special cases, emerging issues, and challenges
Stony soils
When calculating the C and nutrient content in stony soils, the stone volume (stones > 2 mm; Novák et al., 2011) of the soil should be taken into account, as the volume occupied by stone is assumed not to contain a significant amount of C (Rytter, 2012). The correction is generally made with the ‘quantitative pit method’ (Vadeboncoeur et al., 2012), where the rock fragment density is estimated by digging a pit with dimensions large enough to obtain a representative sample of the soil (< 2 mm) and stone (> 2 mm) volume. Subsequently, the volume of the pit and stones are measured. For the pit volume, the pit is lined with plastic and the volume of water necessary to fill it up to the soil surface is tracked (Vadeboncoeur et al., 2012; Mehler et al., 2014; Beem-Miller et al., 2016). The volume of stones is estimated by the water displacement method or by using a hydrostatic scale (Mehler et al., 2014).
In cases when the quantitative pit method is unsuitable (e.g. due to its destructive, labour-intensive or costly nature), other, but less accurate methods can be used. The most common alternatives are the hammer, hydraulic push, and rotary coring methods (Beem-Miller et al., 2016).
Coarse roots
In soils containing a substantial amount of large roots, the same technique can be used as in stony soils (see above) to exclude the root volume from the soil volume (Vadeboncoeur et al., 2012; Mehler et al., 2014). In soils with small roots (e.g. grasslands), the root volume is generally neglected.
Carbonate-containing soil
Soils on calcareous bedrock contain inorganic C in the form of carbonates (e.g. calcite or dolomite). Methods assessing soil C, such as dry combustion or wet oxidation, provide a bulk measure of total soil C consisting of organic and inorganic C components. In most climate-manipulation experiments the primary interest is on how organic C is affected. Therefore, in order to assess effects on organic C, inorganic soil C content needs to be subtracted from total soil C. Under specific conditions, climate manipulations could not only affect organic C, but also directly affect the dissolution or formation of soil carbonates (e.g. their pedogenic formation due to precipitation manipulation in arid or semi-arid environments).
Carbonate contents (and consequently their share in organic C) can be assessed in several ways. Most approaches are based on the comparison of a soil sample containing organic + inorganic C (= total soil C) with a sample free of inorganic C. The technically easiest way of assessing the carbonate content is to treat soil samples with HCl and then conduct a volumetric determination of the released CO2 (Nelson & Sommers, 1982). This gasometric approach is called the ‘Scheibler’ or calcimeter method. The calcimeter method is straightforward, albeit time and labour-consuming, because only a limited number of samples can be treated simultaneously. To assess the carbonate content of a higher number of samples simultaneously, acid washing (e.g. Midwood & Boutton, 1998; Schnecker et al., 2016) or acid fumigation (Harris et al., 2001; Walthert et al., 2010) can be applied to remove carbonates from the samples. These techniques have the advantage that the soil samples can also be used for further analyses (e.g. for isotope measurements). While acid washing can lead to loss of acid-soluble organic C, acid fumigation has been successfully used with calcite (Harris et al., 2001) and even dolomite (Walthert et al., 2010) without solute C loss. Beside these methods, carbonate content can also be assessed non-destructively by FT-IR spectroscopy (Tatzber et al., 2007) or near-infrared analysis (NIRA) (Ben-Dor & Banin, 1990). Such methods are, however, comparatively sophisticated and may be applied only when a detailed characterisation of organic C is foreseen anyway. The thermal gradient (ThG) method has been shown to perform well, especially with regard to the assessment of dolomite contents (Vuong et al., 2016).
2.2.4.3 References
Theory, significance, and large datasets
Davidson & Janssens (2006), Kirschbaum (1995), Melillo et al. (2002), Scharlemann et al. (2014), Tian et al. (2015)
More on methods and existing protocols
Allen et al. (2010), Jobbágy & Jackson, (2000), Maillard et al. (2017), Senesi & Senesi (2016); Vadeboncoeur et al. (2012)
All references
Allen, D. E., Pringle, M. J., Page, K. L., & Dalal, R. C. (2010). A review of sampling designs for the measurement of soil organic carbon in Australian grazing lands. The Rangeland Journal, 32(2), 227-246.
Beem-Miller, J. P., Kong, A. Y., Ogle, S., & Wolfe, D. (2016). Sampling for soil carbon stock assessment in rocky agricultural soils. Soil Science Society of America Journal, 80(5), 1411-1423.
Ben-Dor, E., & Banin, A. (1990). Near-infrared reflectance analysis of carbonate concentration in soils. Applied Spectroscopy, 44(6), 1064-1069.
Chapin, F. S., Matson, P. A., & Vitousek, P. (2011). Principles of Terrestrial Ecosystem Ecology. Springer Science & Business Media.
Davidson, E. A., & Janssens, I. A. (2006). Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature, 440(7081), 165-173.
Gísladóttir, G., Erlendsson, E., Lal, R., & Bigham, J. (2010). Erosional effects on terrestrial resources over the last millennium in Reykjanes, Southwest Iceland. Quaternary Research, 73(1), 20-32.
Guo, L. B., & Gifford, R. M. (2002). Soil carbon stocks and land use change: a meta analysis. Global Change Biology, 8(4), 345-360.
Harris, D., Horwáth, W. R., & van Kessel, C. (2001). Acid fumigation of soils to remove carbonates prior to total organic carbon or carbon-13 isotopic analysis. Soil Science Society of America Journal, 65(6), 1853-1856.
Hobbie, S. E., Nadelhoffer, K. J., & Högberg, P. (2002). A synthesis: the role of nutrients as constraints on carbon balances in boreal and arctic regions. Plant and Soil, 242(1), 163-170.
Jobbágy, E. G., & Jackson, R. B. (2000). The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecological Applications, 10(2), 423-436.
Kirschbaum, M. U. (1995). The temperature dependence of soil organic matter decomposition, and the effect of global warming on soil organic C storage. Soil Biology and Biochemistry, 27(6), 753-760.
Laurel, D., & Wohl, E. (2019). The persistence of beaver‐induced geomorphic heterogeneity and organic carbon stock in river corridors. Earth Surface Processes and Landforms, 44(1), 342-353.
Li, L. J., Zeng, D. H., Yu, Z. Y., Fan, Z. P., Yang, D., & Liu, Y. X. (2011). Impact of litter quality and soil nutrient availability on leaf decomposition rate in a semi-arid grassland of Northeast China. Journal of Arid Environments, 75(9), 787-792.
Maaroufi, N. I., Nordin, A., Hasselquist, N. J., Bach, L. H., Palmqvist, K., & Gundale, M. J. (2015). Anthropogenic nitrogen deposition enhances carbon sequestration in boreal soils. Global Change Biology, 21(8), 3169-3180.
Maillard, É., McConkey, B. G., & Angers, D. A. (2017). Increased uncertainty in soil carbon stock measurement with spatial scale and sampling profile depth in world grasslands: A systematic analysis. Agriculture, Ecosystems & Environment, 236, 268-276.
Matejovic, I. (1997). Determination of carbon and nitrogen in samples of various soils by the dry combustion. Communications in Soil Science & Plant Analysis, 28(17-18), 1499-1511.
Mehler, K., Schöning, I., & Berli, M. (2014). The importance of rock fragment density for the calculation of soil bulk density and soil organic carbon stocks. Soil Science Society of America Journal, 78(4), 1186-1191.
Melillo, J. M., Steudler, P. A., Aber, J. D., Newkirk, K., Lux, H., Bowles, F. P., … Morrisseau, S. (2002). Soil warming and carbon-cycle feedbacks to the climate system. Science, 298(5601), 2173-2176.
Midwood, A. J., & Boutton, T. W. (1998). Soil carbonate decomposition by acid has little effect on δ13C of organic matter. Soil Biology and Biochemistry, 30(10), 1301-1307.
Nelson, D. W., & Sommers, L. (1982). Total carbon, organic carbon, and organic matter. In A. L. Page, R. H. Miller, & D. R. Keeney (Eds.), Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties. (pp. 539-579). American Society of Agronomy, Soil Science Society of America.
Novák, V., Kňava, K., & Šimůnek, J. (2011). Determining the influence of stones on hydraulic conductivity of saturated soils using numerical method. Geoderma, 161(3), 177-181.
Panagos, P., Borrelli, P., Poesen, J., Ballabio, C., Lugato, E., Meusburger, K., … Alewell, C. (2015). The new assessment of soil loss by water erosion in Europe. Environmental Science & Policy, 54, 438-447.
Rytter, R. M. (2012). Stone and gravel contents of arable soils influence estimates of C and N stocks. Catena, 95, 153-159.
Saynes, V., Hidalgo, C., Etchevers, J. D., & Campo, J. E. (2005). Soil C and N dynamics in primary and secondary seasonally dry tropical forests in Mexico. Applied Soil Ecology, 29(3), 282-289.
Scharlemann, J. P., Tanner, E. V., Hiederer, R., & Kapos, V. (2014). Global soil carbon: understanding and managing the largest terrestrial carbon pool. Carbon Management, 5(1), 81-91.
Schnecker, J., Borken, W., Schindlbacher, A., & Wanek, W. (2016). Little effects on soil organic matter chemistry of density fractions after seven years of forest soil warming. Soil Biology and Biochemistry, 103, 300-307.
Senesi, G. S., & Senesi, N. (2016). Laser-induced breakdown spectroscopy (LIBS) to measure quantitatively soil carbon with emphasis on soil organic carbon. A review. Analytica Chimica Acta, 938, 7-17.
Soon, Y. K., & Abboud, S. (1991). A comparison of some methods for soil organic carbon determination. Communications in Soil Science & Plant Analysis, 22(9-10), 943-954.
Sterner, R. W., & Elser, J. J. (2002). Ecological Stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press.
Tatzber, M., Stemmer, M., Spiegel, H., Katzlberger, C., Haberhauer, G., & Gerzabek, M. H. (2007). An alternative method to measure carbonate in soils by FT-IR spectroscopy. Environmental Chemistry Letters, 5(1), 9-12.
Tian, H., Lu, C., Yang, J., Banger, K., Huntzinger, D. N., Schwalm, C. R., … Huang, M. (2015). Global patterns and controls of soil organic carbon dynamics as simulated by multiple terrestrial biosphere models: Current status and future directions. Global Biogeochemical Cycles, 29(6), 775-792.
Vadeboncoeur, M. A., Hamburg, S. P., Blum, J. D., Pennino, M. J., Yanai, R. D., & Johnson, C. E. (2012). The quantitative soil pit method for measuring belowground carbon and nitrogen stocks. Soil Science Society of America Journal, 76(6), 2241-2255.
Vuong, X. T., Prietzel, J., & Heitkamp, F. (2016). Measurement of organic and inorganic carbon in dolomite‐containing samples. Soil Use and Management, 32(1), 53-59.
Wallinga, I., Vark, V. W., Houba, V. J. G., & Lee, J. J. (1989). Plant Analysis Procedures (soil and plant analysis, part 7). Wageningen: Department of Soil Science and Plant Nutrition, Wageningen Agricultural University.
Walthert, L., Graf, U., Kammer, A., Luster, J., Pezzotta, D., Zimmermann, S., & Hagedorn, F. (2010). Determination of organic and inorganic carbon, δ13C, and nitrogen in soils containing carbonates after acid fumigation with HCl. Journal of Plant Nutrition and Soil Science, 173(2), 207-216.
Authors: Leblans NIW1, Stuart-Haëntjens E2, Schindlbacher A3, Vicca S1
Reviewer: Almagro M4
Affiliations
1 Centre of Excellence PLECO (Plants and Ecosystems), Biology Department, University of Antwerp, Wilrijk, Belgium
2 Climate Impacts Research Centre, Department of Ecology and Environmental Science, Umeå University, Abisko, Sweden
2 Department of Biology, Virginia Commonwealth University, Richmond, USA
3 Department of Forest Ecology and Soils, Federal Research and Training Centre for Forests, Natural Hazards and Landscape, Vienna, Austria
4 BC3-Basque Centre for Climate Change, University of the Basque Country, Leioa, Spain