Authors: Zinnert J1, Wood LK1, Johnson DM2, Estiarte3,4
Reviewer: Dickman LT5
Measurement unit: MPa; Measurement scale: leaf or stem; Equipment costs: €€; Running costs: €; Installation effort: medium; Maintenance effort: low; Knowledge need: medium; Measurement mode: manual
Plant water relations are important in the maintenance of turgor and control the hydration of cells for proper physiological functioning, especially during periods of drought. As plants lose turgor, there is a decline in critical physiological functions such as cell expansion and photosynthesis. Water potential is an indicator of plant water status and is made up of both osmotic and turgor potential. Pressure volume curves describe the relationship between bulk water potential (Ψw, movement driven by a pressure gradient) and relative water content (RWC) through iterative measurements of a drying leaf. From the mass and water potential measurements several important parameters can be derived: turgor loss point (TLP), bulk modulus of elasticity (ε), and osmotic potential (Ψo). TLP indicates the point at which the turgor pressure in cells is zero and can be used as an indicator of tolerance of leaves to drought stress. ε indicates plant cell-wall elasticity (for more detail on ε see Brartlett et al. 2012). Ψo is the pressure due to solutes in living cells within the leaves. Pressure-volume curves can be used in coordination with stomatal conductance (gs), stem- or leaf-specific hydraulic conductivity (KS, KL), or vulnerability to embolism (described in Pérez-Harguindeguy et al. 2013) to gain further insight into hydraulic functioning within the plant.
Across plant species, there is a continuum of responses to drought that result in different ranges of water potentials. The most conservative end of the spectrum, isohydry, is characterised by stomatal closure at “set-point” water potentials, or potentially by having constant access to water (i.e. deep roots. Isohydric species prevent the plant from experiencing embolism-inducing xylem pressures and reducing the need for solute accumulation/osmostic adjustment to maintain turgor as soil dries (Meinzer et al., 2014). The opposite end of the spectrum, anisohydry, encompasses plants that do not regulate water potential to a set value or those that may experience dry soils in their rooting areas. Water potentiasl decline in more anisohydric species as the soil dries. (Tardieu & Simonneau, 1998; McDowell et al., 2008; Meinzer et al., 2014). TLP and Ψo derived from pressure-volume curves provide insight on the isohydric and anisohydric response to water stress (Tyree & Hammel, 1972; Meinzer et al., 2016). TLP, ε and Ψo are traits that are mechanistically linked to drought tolerance and can be used to determine the degree to which plants resist or adapt to temporary or prolonged water stress, enhancing our ability to make predictions about how climate change will affect plant success and thus a useful measurement in climate-change studies.
5.9.1 What and how to measure?
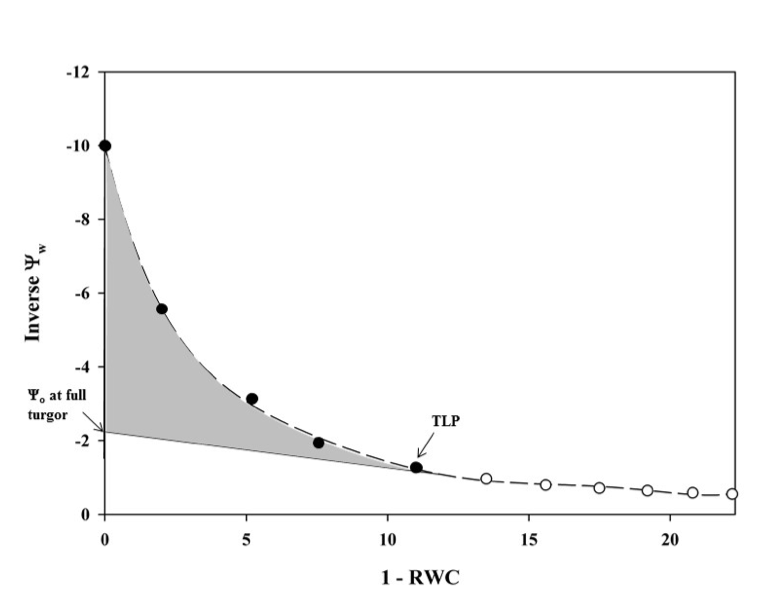
Pressure volume curves (Figure 5.9.1.1) are generated by plotting the inverse of the negative Ψw against the relative water deficit (100 – RWC) obtained using a balance and a Scholander pressure chamber (described below). The resulting figure has two distinct parts: the non-linear portion and the linear portion of the plot (Figure 5.9.1.1). Leaves or distal ends of shoots from whole plants should be excised pre-dawn or from well-irrigated plants to ensure starting the measurements at the highest Ψw or turgid weight (Tyree & Hammel, 1972). It is best to limit stomatal water loss from samples between collection and processing. This can be done by wrapping the sample in plastic wrap, inserting the sample in a sealed plastic bag with a damp paper towel, and placing the bag in a cooler to decrease respiration and water loss. For optimal measurements, samples should be processed as close to collection as possible. Each sample is weighed before and after obtaining the water potential using a Scholander pressure chamber which is described in detail in Pérez-Harguindeguy et al. (2013). It should be noted that samples should be bagged when not bench dehydrating and should be kept in pastic bags for 20 minutesd before making any water potential measurements (i.e. after drying on the bench, place samples in plastic bags prior to making water potential measurements). The sample is placed in the gasket with the stem pointing upward and a positive pressure is applied gradually until water appears on the cut end of the sample. The positive pressure applied is equal to the negative pressure within the stem (Pérez-Harguindeguy et al., 2013). The sample is laid on the bench to desiccate and the measurements are repeated. Subsequent measurements are taken every 2–5 minutes initially, depending on the humidity of the room and the speed with which the sample loses water. After the sample has lost some water (differs depending on species), sample intervals can be increased to 15 minutes to 2 hours. Measurements are taken until 6–8 points have been collected along the linear portion of the pressure volume curve. It may take some experimentation to determine how long it takes to reach this point. Relative water content is calculated as:
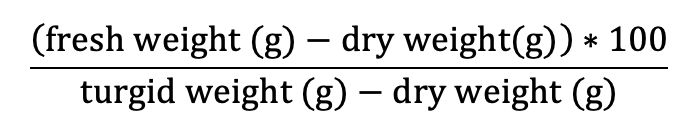
where the fresh weight is the mean of the weights taken before and after each Ψw iterative measurement, the turgid weight is the weight of the sample taken before the first measurement, and the dry weight is the weight of the dried sample taken after all measurements are completed. Leaves should be dried at a maximum of70 °C for 72 h to constant weight (Pérez-Harguindeguy et al., 2013) to determine dry weight. The temperature and time for drying depend on the study question, how many samples are dried, the size, thickness and type of the plant material (e.g. large, fleshy or succulent leaves need more time) see protocol 2.1.1 Aboveground plant biomass for more details on the drying.The x-axis of the plotted data is 100 – RWC and the y-axis is the 1 / -Ψw at each weight (Figure 5.9.1.1). From this graph, TLP and Ψo at TLP can be estimated from the inflection point where the curved portion of the line meets the linear portion. The linear portion can be extrapolated to the y-intercept, which will give the Ψo at full turgor pressure.
The extrapolated linear portion of the line can be subtracted from the curve to get the turgor potential (Ψp) using the equation Ψw = Ψo + Ψp. To calculate ε, Ψp can be plotted separately with RWC on the x-axis and a non-linear curve fitted to the line, where ε is the derivative of that curve:
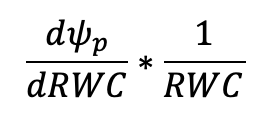
The Scholander pressure chamber is a useful instrument for investigating plant water status by applying positive pressure to equal the tension of the water within the xylem. To operate, a compressed nitrogen tank needs to be connected to the intake valve on the pressure chamber. While acquiring samples, the control valve on the chamber should be set to “off”. Slowly open the valve on the nitrogen tank. For the initial measurement, a fresh excised sample is cut with a sharp blade to ensure a clean surface. The sample is placed through the rubber gasket on the lid of the chamber with the cut edge facing upward. Put the lid back on the chamber by pushing down and turning clockwise to stop. This seals the chamber with fresh foliage inside. Turn the control valve to “chamber” to pressurise the chamber. The rate valve should be set so that the pressure increases at the desired rate (this should be no more than 0.1 bar per second). Once the rate valve is set for the desired pressure increase per time, it should not be adjusted. Using the rate valve as an on/off valve will result in valve failure. As the positive pressure approaches the negative pressure within the plant sample, a droplet of water will appear on the cut edge of the sample and the gas should be cut off without venting the chamber. The pressure (in bars or MPa) can be read on the pressure gauge. Once the pressure is recorded, turn the control valve to “exhaust” to release pressure from the chamber. Remove the lid, rubber gasket, and sample. For more detailed information see Pérez-Harguindeguy et al. (2013).
Where to start
Barlett et al. (2012), Meinzer et al. (2014, 2016), Pérez-Harguindeguy et al. (2013), Tardieu & Simonneau (1998), Tyree & Hammel (1972)
5.9.2 Special cases, emerging issues, and challenges
Time of drying depends on cuticular resistance, specific leaf area of sample, and relative humidity of the room in which the measurements are being performed. Pressure-volume curves of similar species can be acquired from previous publications or multiple trials can be done to look at speed of water loss and associated Ψw.
Bulk modulus of elasticity and Ψo at TLP may be underestimated by pressure volume curves constructed as above (Cheung et al., 1976). There can be accuracy issues with artificial rehydration of samples in the lab: rehydration can result in rapid changes in Ψo and TLP leading to values that are not representative of the actual status of plants under study (Meinzer et al., 2014). This presents issues when assessing samples in drought treatments and comparisons should be limited to sample differences relative to each other within that experiment rather than values obtained in other studies.
5.9.3 References
Theory, significance, and large datasets
Barlett et al. (2012), Cheung et al. (1976), Meinzer et al. (2016), Tardieu & Simonneau (1998), Tyree & Hammel (1972)
More on methods and existing protocols
Meinzer et al. (2014), Pérez-Harguindeguy et al. (2013)
All references
Bartlett, M.K., Scoffoni, C. & Sack, L. (2012). The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: a global meta‐analysis. Ecology Letters, 15(5), 393-405.
Cheung, Y.N.S., Tyree, M.T., & Dainty, J. (1976). Some possible sources of error in determining bulk elastic moduli. Journal of Experimental Botany, 23(74), 267-282.
McDowell, N., Pockman, W. T. Allen, C. D., Breshears, D. D., Cobb, N., Kolb, T., … Yepez, E. A. (2008). Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytologist, 178, 719-739.
Meinzer, F. C., Woodruff, D. R., Marias, D. E., McCulloh, K. A., & Sevanto, S. (2014). Dynamics of leaf water relations components in co-occurring iso- and anisohydric conifer species. Plant, Cell & Environment, 37(11), 2577-2586.
Meinzer, F. C., Woodruff, D. R., Marias, D. E., Smith, D. D., McCulloh, K. A., Howard, A. R., & Magedman, A. L. (2016). Mapping ‘hydroscapes’ along iso- to anisohydric continuum of stomatal regulation of plant water status. Ecology Letters, 19, 1343-1352.
Pérez-Harguindeguy, N., Díaz S., Garnier, E., Lavorel, S., Poorter, H., Jaureguiberry, P., … Cornelissen, J. H. C. (2013). New handbook for standardized measurement of plant functional traits worldwide. Australian Journal of Botany, 61, 167-234.
Tardieu, F. & Simonneau, T. (1998). Variability among species of stomatal control under fluctuating soil water status and evaporative demand: modelling isohydric and anisohydric behaviours. Journal of Experimental Botany, 49(Special Issue), 419-432.
Tyree, M.T. & Hammel, H.T. (1972). The measurement of turgor pressure and water relations of plants by the pressure-bomb technique. Journal of Experimental Botany, 23(74), 267-282.
Authors: Zinnert J1, Wood LK1, Johnson DM2, Estiarte3,4
Reviewer: Dickman T5
Affiliations
1 Department of Biology, Virginia Commonwealth University, Richmond, USA
2 Warnell School of Forestry and Natural Resources, University of Georgia, Athens, USA
3 CSIC, Global Ecology Unit CREAF-CSIC-UAB, Bellaterra, Spain
4 CREAF, Cerdanyola del Vallès, Spain
5 Earth and Environmental Sciences Division, Los Alamos National Laboratory, Los Alamos, USA